Top 5 Reasons Emerging and Early-Stage Biopharma Should Consider the Japan Market
For emerging biopharma and early-stage pharmaceutical companies, global strategy is no longer optional—it is a core value driver of valuation, capital efficiency, and long-term risk diversification. Japan, the world’s third-largest pharmaceutical market, represents a significant strategic opportunity within global development planning.
Here are five strategic reasons to consider Japan early in your development plan.
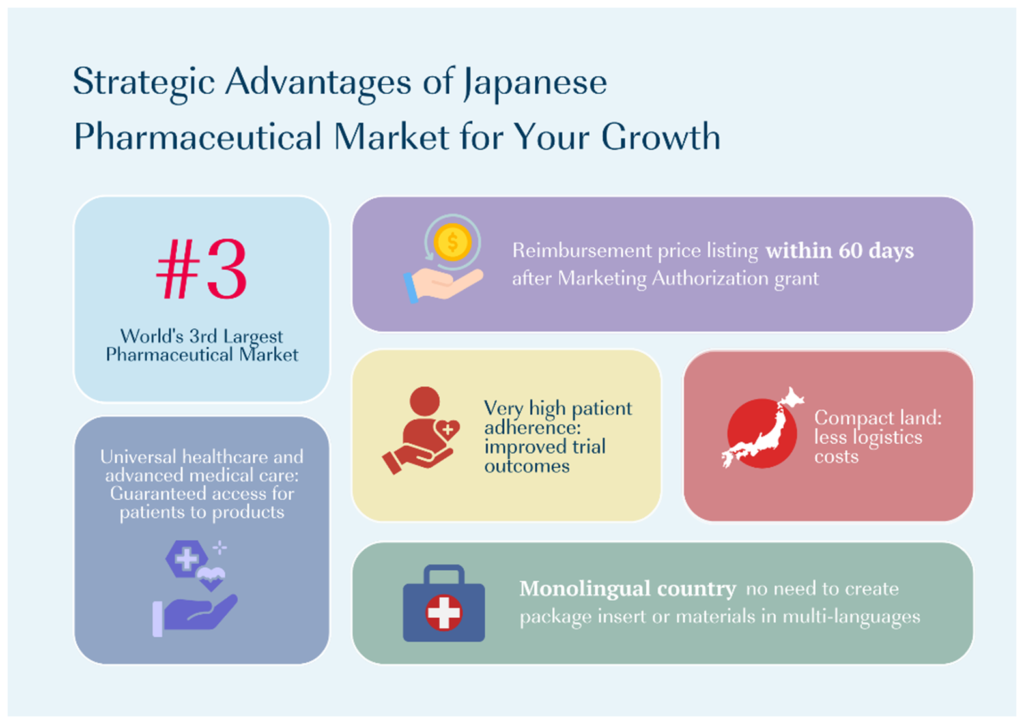
1. The World’s Third-Largest Pharmaceutical Market
Japan represents one of the largest pharmaceutical markets globally, supported by universal healthcare coverage and stable reimbursement under the National Health Insurance (NHI) system.
For emerging companies, this means:
- Meaningful revenue potential outside the U.S.
- Predictable pricing and reimbursement pathways
- Strong uptake of innovative therapies, particularly in oncology and rare disease
- High per‑patient spend supported by public insurance
- Faster post‑approval uptake compared with fragmented EU markets
Japan offers scale with structural stability—an increasingly valuable combination.
2. Regulatory Pathways That Support Innovation
Japan’s PMDA is highly aligned with ICH standards and increasingly collaborative with global regulators.
Key advantages include:
- Acceptance of global clinical data, allowing developers to avoid duplicative, Japan‑only clinical programs when planned appropriately
- Expedited pathways such as Sakigake designation
- Conditional approval systems for regenerative medicine and rare diseases
When integrated early into global development planning, Japan can be included efficiently rather than treated as a delayed market entry.
3. A Rapidly Aging Population Driving Structural Demand
Japan has the most rapidly aging population among developed nations:
- Roughly 30% of the population is aged 65+1
- High prevalence of oncology, cardiovascular, metabolic, and neurodegenerative diseases
- Growing demand for cell & gene therapy, rare disease treatments, and precision medicine
Demographics in Japan are not cyclical—they are structural.
For companies focused on chronic disease, oncology, CNS, or specialty therapeutics, Japan represents a durable, long-term demand driver. This is particularly compelling for emerging biopharma building assets in age-associated indications.
4. High-Value Licensing and Partnership Opportunities
Japan has a mature ecosystem of pharmaceutical companies actively seeking innovative external assets to fill future portfolio gaps. Strong development and commercialization capabilities of Japan pharma enable faster local execution.
For early-stage companies, this creates:
- Attractive regional licensing deals with meaningful upfronts and milestones
- Strategic co-development partnerships
- Risk-sharing commercialization models
Importantly, Japanese pharmaceutical companies often engage earlier in the asset lifecycle than their U.S. or EU counterparts, taking a long-term portfolio-driven view rather than a purely transactional approach.
Japan rights can also serve as a non-dilutive financing mechanism—an important consideration for venture-backed companies.
5. Portfolio and Valuation Enhancement
From an investor’s perspective, Japan integration enhances both upside potential and capital efficiency. Integrating Japan into early development strategy can materially impact enterprise value:
- Broader global market potential strengthens investor positioning
- Japan as a gateway into other APAC countries with accelerated approvals
- Regional licensing provides capital without equity dilution
- Parallel development can accelerate global peak revenue timelines
Japan is not merely an expansion market—it is a strategic lever.
Strategic Bottom Line
Japan should not be viewed as a secondary or post-U.S./EU market—but as an integral part of early global development strategy. For emerging and early-stage biopharma companies, proactive Japan strategy—whether through direct development, regional partnership, or hybrid models—can:
- Increase asset value
- Improve capital efficiency
- De-risk global commercialization
- Accelerate revenue diversification
Delayed Japan entry often leads to:
- Redundant local trials
- Misaligned endpoints for PMDA
- Lost partnering leverage
CMIC Group supports sponsors with strategic consulting and end-to-end Japan market entry solutions, helping translate opportunity into executable, value-driven market access strategies. With deep expertise spanning regulatory strategy, clinical development, CMC, and commercialization, CMIC enables sponsors to design Japan strategy proactively—rather than reactively.
Begin your journey in Japan today:
Contact our Consulting Team https://en.cmicgroup.com/solutions/specialized-services/japan-market-entry/
Sources:
- “Japan’s Aging Society as a Technological Opportunity” https://carnegieendowment.org/research/2024/10/japans-aging-society-as-a-technological-opportunity
